, Master Your Oxytocin
This might sound like a strange claim, so bear with me. Earlier in the book, I explained how the Korean proverb "When your cousin buys land, your stomach hurts" turns out to be more physiologically literal than anyone expected. Now, one of the hottest topics in science right now is the gut microbiome — the vast population of bacteria living inside your intestines. These days, the term microbiome comes up everywhere. If your body is made up of roughly 30 trillion human cells, the bacteria in your gut number approximately 38 trillion. More bacterial cells live in your intestines than human cells make up your entire body. And those 38 trillion bacteria are not all the same — they comprise about 1,000 distinct species, a staggering diversity. Analyze their collective genome and you find roughly 100 to 150 times more genes than exist in the entire human genome. It raises a genuinely disorienting question: do I live inside these bacteria, or do they live inside me? Together, they weigh a mere 200 grams. So why has this tiny population sent the global scientific community into such excitement? Let us take a look.
The Shocking Truth from Fat Microbiomes
My own interest in the microbiome started with media coverage of the so-called "obesity bacteria." A landmark 2013 study published in Science tackled one of the most persistent puzzles in metabolism: why, among genetically identical twins, one can be obese while the other stays thin. The answer was startlingly simple. Researchers transplanted microbiome samples from the feces of an obese twin and a lean twin into laboratory mice. Mice that received the obese twin's microbiome got fat. Those that received the lean twin's stayed slim. When I first read this, I thought: "Come on, that can't be right." Body composition is shaped by so many factors that I assumed the researchers had failed to control for something.1
But when I read the paper more carefully, I found that the experimental conditions were meticulous. And the follow-up experiment was even more interesting. The researchers housed the fat-microbiome mice and the lean-microbiome mice in the same cage, and the fat mice started getting thinner. "Wait — losing weight can be this easy?" Reading further, the mechanism revealed itself with an almost comic simplicity. Lab mice do not have designated bathrooms. They defecate wherever they happen to be, step in each other's droppings, and — being mice — eat them too. Through this indelicate process, the lean mice's microbiome migrated into the fat mice's guts. If this principle could be applied commercially, the multi-billion-dollar diet industry might find itself turned on its head.
Autism and the Microbiome
But even more compelling than the obesity research was the emerging connection between the microbiome and autism. Many studies have looked at whether antibiotic use is linked to autism, but the field is split right down the middle, with no consensus in sight. To me, this is entirely predictable. Autism has both genetic and environmental causes, and untangling them is extremely difficult.*
The hallmark of autism spectrum disorder is impaired social functioning. As I discussed earlier, individuals with autism often struggle to maintain eye contact, have trouble reading emotions from facial expressions, and even when they pick up on an emotion, find it hard to share in it. Given what we know about oxytocin and autism — low oxytocin levels and mutations in the oxytocin receptor are both linked to reduced empathy and higher autism risk — the gut connection becomes relevant. Multiple genes governing the oxytocin receptor have been identified, and the number of variants an individual carries can help predict the severity of symptoms.
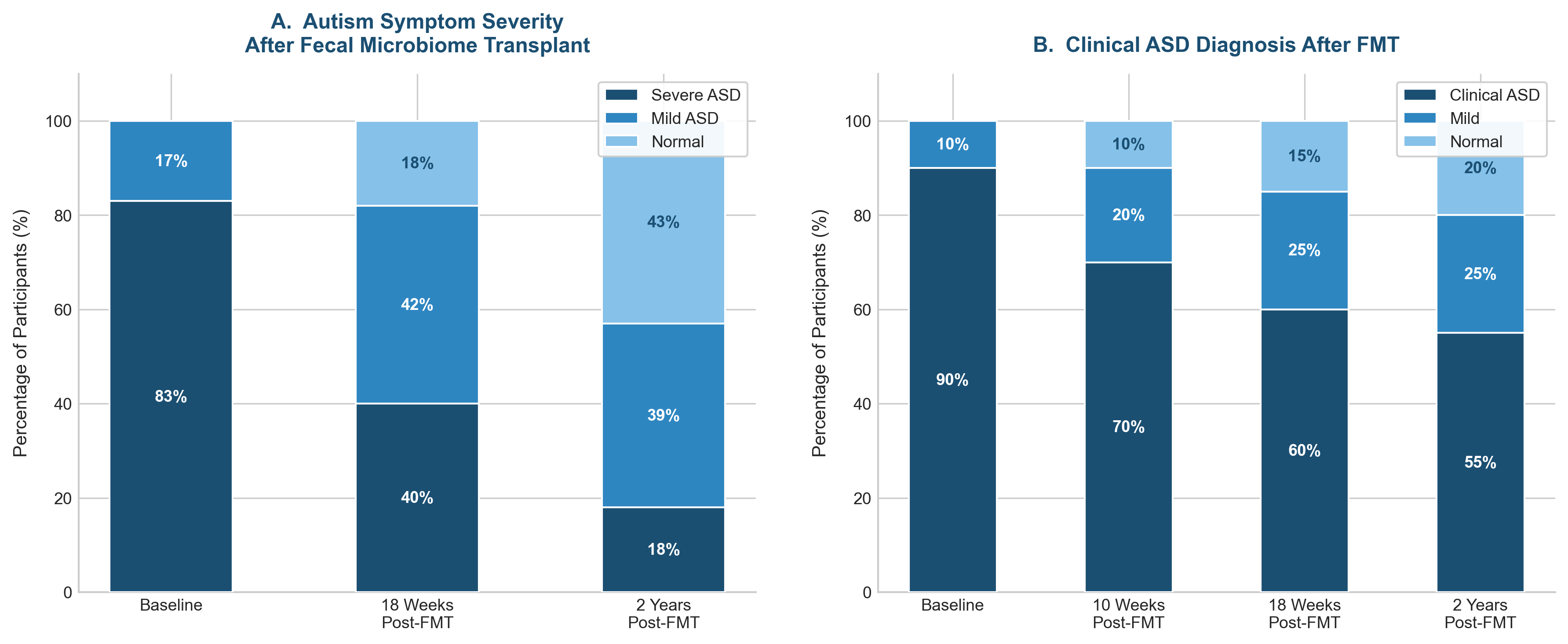
So why might antibiotics matter? Because they devastate the microbiome. About 70 percent of people with autism spectrum disorder have gastrointestinal problems. A research team in Arizona tried something ambitious: they first treated children with autism with a two-week course of antibiotics, then transplanted healthy microbiome samples over seven to eight weeks and observed the results.
The results were, in a word, stunning. Gut symptoms improved by roughly 80 percent, and autism symptoms showed meaningful improvement as well. Two years later, the same team published follow-up data. Of the 18 children in the study, a significant number had either lost their autism symptoms entirely or seen them become quite mild. Fecal analysis of 16 of the 18 participants revealed substantial increases in Bifidobacterium and Prevotella — two types of beneficial gut bacteria. The implication is remarkable: changing the gut microbiome alone produced dramatic improvements in both digestive health and social behavior in children with autism.2
\* Genetic factors explain only about 10--20 percent of autism spectrum disorder. When autism arises from environmental or acquired causes, genetic predisposition acts as a trigger. For example, if an individual already carries many autism-related genetic variants, their condition cannot be attributed solely to antibiotic use. Conversely, in the rare case where no such genetic variants are present, antibiotics alone might theoretically trigger symptoms — but this has not been scientifically confirmed.
Change Your Life to Master Your Gut
A natural question follows: if microbiome changes can improve social functioning in children with autism, could they help the rest of us too? Some Korean university hospitals are already investigating this through clinical trials. But we do not all need fecal transplants. We can reshape our gut ecology through what we eat three times a day.
The foods that support a healthy microbiome are refreshingly ordinary. Gut bacteria feed primarily on carbohydrates — specifically, the kind that survive the small intestine and reach the colon intact. Those are vegetables and dietary fiber. This is exactly why eating more vegetables matters. Second, cut down on processed food. Your gut is home to both beneficial and harmful bacteria, and keeping them in balance is critical. Processed foods starve the good bacteria and feed the bad, because they contain virtually no fiber. A diet heavy in processed food and sugar destabilizes the microbial ecosystem, potentially fueling chronic inflammation and weakening the immune system.
Third, improve your overall lifestyle. Regular exercise, moderate drinking, no smoking, enough sleep, and healthy weight management all contribute to a healthier gut. For exercise, try to include activities that get you genuinely breathless — brisk walking, cycling, resistance training. Choose exercises appropriate for your joints, and for oxytocin's sake, exercise with others whenever possible. Alcohol and smoking are terrible for gut health. This is so obvious that I will not belabor the point.
Adequate sleep and time-restricted eating are also essential. These two deserve special emphasis because irregular eating habits — particularly the Korean culture of yasik (late-night snacking) — do real damage to both gut health and sleep quality. Consider the chemical formula for glucose: C~6~H~12~O~6~. Glucose is basically carbon atoms with water molecules attached. If you wake up in the middle of the night after a carb-heavy late snack, it is because the water released during carbohydrate metabolism needs to come out. And carbohydrates raise blood insulin, which promotes wakefulness and disrupts your sleep cycle. Is protein a better late-night option? Not really — protein demands much more digestive energy, forcing the stomach and intestines to keep working when they should be resting. A heavy protein snack before bed may actually be worse.3
How the Microbiome and Oxytocin Work Together
The way the microbiome and oxytocin affect social behavior follows remarkably parallel tracks. When antibiotics wipe out gut bacteria, oxytocin levels drop right along with them, and sociability declines. Among beneficial gut bacteria, Lactobacillus reuteri stands out: it not only stimulates oxytocin production but also boosts social behavior. And the overlap between what L. reuteri does and what oxytocin does is almost eerie — anti-cancer, anti-inflammatory, antidepressant, immune-boosting, anti-obesity, wound-healing. The list is nearly identical.
So what happens when you give Lactobacillus reuteri to animals that lack functional oxytocin receptors? The results are revealing. Without working oxytocin, Lactobacillus's wound-healing, immune, and anti-obesity effects drop sharply — or disappear altogether. And when the vagus nerve, the main highway of the parasympathetic nervous system, is severed, both oxytocin's and Lactobacillus's beneficial effects collapse in tandem. These findings fit neatly with recent research showing that stimulating the parasympathetic nervous system through meditation, exercise, or even electrical stimulation can improve depression and a range of other mental health conditions. The conclusion is hard to avoid: master your gut, and you master your oxytocin. That is not an exaggeration. It is physiology.4